Abbott has received an FDA emergency use authorization (EUA) for its Abbott RealTime SARS-CoV-2 molecular test to detect COVID-19.
The company will immediately ship 150,000 tests — which are used on the Abbott m2000 RealTime system — to existing US customers.
The company said it will ramp up production at its US manufacturing location to the highest levels possible, with the goal of providing up to 1 million tests per week.
“A global challenge like coronavirus requires the commitment and cooperation of everyone who has the ability to help address it,” said Miles D. White, chairman and chief executive officer of Abbott. “I’m proud of the Abbott team and what they’ve accomplished in such a short period of time, and I want to thank the administration and FDA for their partnership in making this happen.”
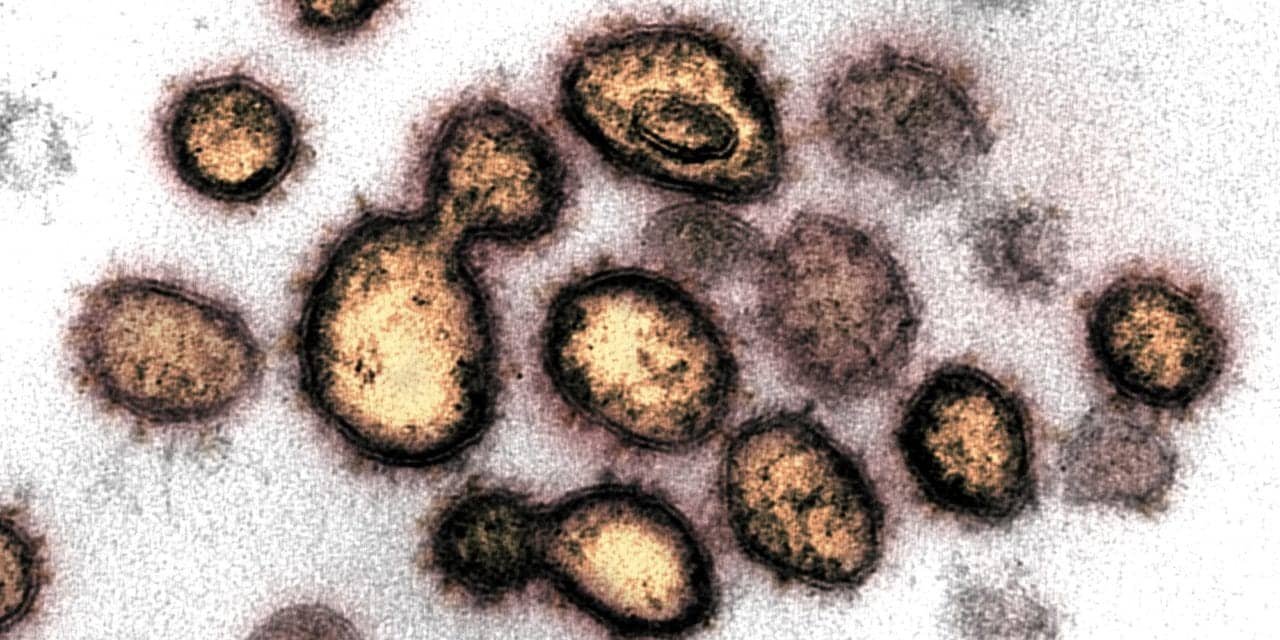
Image credit: NIAID-RML. Novel Coronavirus SARS-CoV-2. This transmission electron microscope image shows SARS-CoV-2, the virus that causes COVID-19, isolated from a patient in the US. Virus particles are shown emerging from the surface of cells cultured in the lab. The spikes on the outer edge of the virus particles give coronaviruses their name, crown-like.