Patient monitoring technology like pulse oximetry is providing a safety net for newborns and infants in neonatal ICUs, while new advances in home monitoring look to deliver children’s vital signs to parents’ smart phones.
In today’s healthcare facilities, effective respiratory care goes hand in hand with effective patient monitoring technology. From critical care to postsurgical, or mechanical ventilation patients under anesthesia or opioid analgesics, a consistent safety net is required in order to reduce adverse events and ensure positive patient outcomes. But when it comes to neonatal monitoring, especially in neonatal intensive care units (NICUs), an extra layer of care and safety is necessary, and that layer is often in the form of pulse oximetry.
Pulse oximetry, which measures blood oxygenation and heart rate, is one of the most common respiratory monitoring techniques. Its noninvasive nature makes it ideal for use with babies and infants, and in the NICU at Reading Hospital in West Reading, Pa, it’s a standard of care.
“Anytime we need to initiate any type of oxygen or support, even in the delivery room, per our NRP (Neonatal Resuscitation Program) guidelines, right away the child goes on a pulse oximeter,” said Jennifer Mahone, BS, RRT-NPS, a neonatal/pediatric clinical specialist at Reading Hospital. “Then we will transport the child over to the NICU where they are weighed and immediately hooked up to our monitors, which continuously measure their blood pressure, respiratory rate, blood oxygenation, and more.”
Mahone explained that her NICU, which was created in 1988 and currently serves approximately 600 babies a year, treats newborns and infants suffering from a number of illnesses and disorders, including any complications that come from premature birth. Monitoring these children is much like any other kind of patient monitoring, with a few exceptions. “The mechanics of monitoring are essentially the same [as adults], but we have to be mindful of skin care, especially on our smaller children. We try to be careful with probes and not to have anything too sticky on their skin,” Mahone explained.
For many who work in the realm of neonatal medicine, like Mahone, watching and monitoring infants is a passion. Thankfully, the technology available today has made the process more advanced and more accurate than ever before.
Reading Hospital’s NICU uses IntelliVue monitors by Philips Healthcare and Nellcor pulse oximeters by Covidien. IntelliVue technology offers integrated point-of-care monitoring of ECG, heart rate, respiratory rate, and connectivity to other devices and hospital systems. Nellcor oximetry ensures the child’s blood oxygenation remains within the proper range.
“All of our NICU babies are on pulse oximetry. We have certain parameters based on their age and their weight,” Mahone explained, noting they often use Vermont Oxford Network settings. “We are able to program our saturation parameters into the monitors so we can constantly be titrating their O2. If their saturations are too low or too high, the device will notify us that they need more or less oxygen.”
Working together, these monitoring devices help ensure positive outcomes for Reading’s newborns. “The ultimate goal of our NICU is to save the lives of these most fragile patients. Monitoring technology continues to grow and improve, and helps us to provide that extra layer of safety to our tiny patients,” she said.
The Need for Monitoring
Monitoring newborns and infants is a delicate task, but it’s one that is absolutely vital to a baby’s well-being in the first few minutes of life and, in some instances, beyond. Studies have shown that neonatal and pediatric monitoring not only saves infant lives, but can serve as a preventive strategy for some adult diseases that, unchecked, can develop early in life. For instance, the expanded use of ECG for children “lies largely in the realization that early identification of life-threatening arrhythmogenic disorders, which often manifest in infancy, childhood or even later, may allow initiation of effective preventive therapy,” according to a Task Force Report published in the European Heart Journal.1
In addition, newborn screening using pulse oximetry is helpful for identifying a number of life-threatening conditions, such as critical congenital heart defects (CCHDs), according to the Centers for Disease Control and Prevention (CDC).2 CCHDs are structural heart defects often associated with hypoxemia and other physiologic changes, which can be detected by pulse oximeters.2 As the CDC reports, CCHDs represent an estimated 25% of all congenital heart defects and infants with the disorder are at risk of mortality early in life.2
As a result, states all across the country have been passing or proposing legislation to require CCHD screenings using pulse oximetry. Currently, 29 states have enacted laws and three have added the test to their mandatory newborn screening (NBS) panels statewide.3 In Pennsylvania, the test is a regulatory addition to the state’s NBS Panel, which means delivery facilities like Reading Hospital perform the test for all newborns. “As per policy, all of our newborns at our hospital are screened 24 hours after birth,” Mahone said, adding that she believes the screenings make sense. “If a test as simple as pulse oximetry can detect the possibility of a critical congenital heart defect, it would be crazy not to screen our patients.”
In fact, a 2012 study estimated that universal screening in the United States would identify 248 more cases of CCHD each year and prevent 110 infant deaths annually.4 “I do believe these screenings can save lives,” Mahone said. “Even if we have only one positive result in every several hundred babies, that is one life we have potentially saved. It seems like a no-brainer to me.”
The Growth of Home Monitoring
Children in the NICU at Reading Hospital are monitored continuously, through discharge. But for those infants who require additional monitoring outside the hospital, Reading makes arrangements for home monitoring devices supplied by an outside company. The transition from clinical to home monitors is seamless, Mahone emphasized, with Reading’s neonatologists continuing follow-ups for as long as children require monitors.
There has been substantial growth in the use of home monitoring devices. According to an April 2012 report by Markets&Markets, a market research company and consulting firm, the patient monitoring systems market was worth $14.2 billion in 2011 and is expected to reach $18.9 billion in 2016.5 Growth “is being driven by the increasing number of geriatric population, rising incidences of lifestyle diseases, home and remote monitoring, and portability of devices,” the report stated.5 The growth and demand have created the opportunity for companies to explore next-generation products. “Companies are forming joint-ventures and collaborations to … create innovative products to suit the needs of the patients,” according to the same report.5
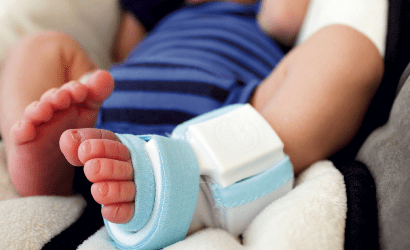
One such innovative home monitoring device being developed is the Vitals Monitor by Owlet Baby Care, Salt Lake City. The monitor is a neonatal/infant “smart sock” with an integrated pulse oximeter designed to fit comfortably onto a child’s foot. The system transmits the baby’s heart rate, oxygen levels, skin temperature, sleep quality, and sleep position (including rollover alerts) to a parent or guardian’s smartphone.
“The Owlet Vitals Monitor is the first ‘wearable technology’ in the infant space, and it’s especially unique because it applies a safe, proven hospital technology in a new way,” said Jordan Monroe, communications manager for Owlet. “Utilizing multiple sensors, the system can grow with the child. Unlike current pulse oximeters, the monitor will continue to work as long as it fits the child’s foot, and it has been beta tested on infants up to two years old.”
The current device, which is not yet FDA-approved, is designed for home monitoring, but the eventual medical use of the Vitals Monitor will be different from the current version. “We’ve actually had a lot of physicians show interest with bringing it into clinical use,” Monroe said, noting that it’s the data-collecting possibilities for the Owlet Vitals Monitor that is most intriguing to neonatal care providers. “There’s a specific amount of data that is collected using pulse oximetry technology when a baby is first born, and we’re working to take that level of information and apply it to our device,” he said.
The Vitals Monitor offers advancements to contemporary systems for measuring pulse oximetry. “Our system offers five sensors, opposed to the industry average of two. With every other pulse oximeter, you can only get two combinations of readings. However, with our smart sock, we’re able to get 9 different combinations. This allows for a substantial jump in data return,” said Monroe, who added that the Owlet team is complying with the FDA process to add an alarm and other necessary features to the Vitals Monitor system in an effort to expand its use.
As the Markets&Markets report on patient monitoring stated: “The future is going to be wireless, non-invasive, and remote monitoring.”5 Owlet, which will begin shipping its first home units in November, is prepared to fill the need for innovation in neonatal monitoring. The company is working with pediatricians and healthcare providers to expand on the next version of the technology, which Monroe believes will take the device to a whole new level. Looking even farther ahead, Owlet wants to be able to send a notification to a parent’s phone that indicates that a child has received less sleep than the average amount. This capability enables parents to be aware of a potentially low immune system and take actions to help prevent sickness, or to become aware of a health-related incident before it occurs.
“We’re in an exciting time for this type of technology,” Monroe said. “It always helps that we can collect and send this type of information through mobile devices, such as a smart phone. I really think as the industry progresses, the collection of information and the utilization of technology will become similarly accessible and beneficial.”
RT
For more information, contact [email protected].
References
- Schwartz PJ, et al. Task Force Report: Guidelines for the interpretation of the neonatal electrocardiogram. European Heart Journal. 2002; 23, 1329–1344. Accessed from: http://eurheartj.oxfordjournals.org/content/23/17/1329.full.pdf
- Screening for Critical Congenital Heart Defects. Centers for Disease Control and Prevention. 2013, Jul. Accessed from: http://www.cdc.gov/ncbddd/pediatricgenetics/pulse.html
- The Newborn Coalition. Accessed from: http://newborncoalition.com.
- Peterson, C. et al. A cost-effectiveness analysis of universal pulse oximetry screening to detect critical congenital heart disease in U.S. newborns. Circ Cardiovasc Qual Outcomes. 2012; 5: A228.
- MarketsandMarkets. Patient monitoring devices market (2011-2016) – global trends, end-user & value-volume analysis. 2012 April. Accessed from: http://www.marketsandmarkets.com/Market-Reports/patient-healthcare-monitoring-systems-devices-market-678.html