Vyaire Medical Inc’s bellavista 1000e ventilator has been granted 510(k) clearance from the US FDA, according to a press announcement from Vyaire. The device debuted at AARC 2019 in New Orleans, along with a suite of Vyaire products. (See images below.)

Side view of Vyaire’s new bellavista 1000e ventilator at AARC 2019.
The device is the newest addition to Vyaire’s high-end ventilation portfolio offering a comprehensive solution for hospitals with the integration of high flow oxygen therapy and advanced synchrony support for neonatal to adult patients in a single device. Clinicians can rapidly change therapies on a broad range of patients in an effortless way.
“We began using bellavista exclusively in 2017, when it was available in Europe. It allows us to ventilate invasive, noninvasive, nasal CPAP and perform high flow oxygen therapy with one device enabling targeted, efficient patient care in any situation” said Vincenzo Cannizzaro, MD, PhD, Deputy Chief Physician of PICU/NICU, University Children’s Hospital, Zurich, Switzerland.
Designed exclusively around enhancing the caregiver’s experience, the bellavista 1000e’s intuitive touch screen provides advanced graphics to easily visualize the patient’s pulmonary status, enhancing situational awareness for the caregiver. Whether its escalating care or weaning a patient efficiently, the bellavista 1000e ventilator simplifies the process by delivering optimal workflows with the goal of reducing operator error and ventilator lengths of stays.
“Clinicians, respiratory therapists and bedside caregivers provided critical input in the design of this ventilator and helped us ensure we addressed their challenges in all patient care circumstances,” said Lisa Rose, Chief Marketing and Innovation Officer, Vyaire Medical. “The bellavista 1000e is a solution that provides the ultimate versatility in therapy and it can be adjusted quickly depending on patient need in time sensitive situations. These differentiators are critical in a healthcare environment which expects innovative products to improve patient outcomes. Vyaire is dedicated to developing the kinds of systems and products that meet the needs of healthcare providers.”
The Vellavista 1000e debuted at AARC 2019, and was on display with Vyaire’s new Airlife oxygen mask, SuperNO2VA anesthesia mask, and its PFT line of technologies: Vyntus One and Vyntus Body with SentrySuite software.

Vyaire’s new Airlife oxygen masks at AARC 2019.

Close up of the delivery port for Vyaire’s new Airlife oxygen mask at AARC 2019.
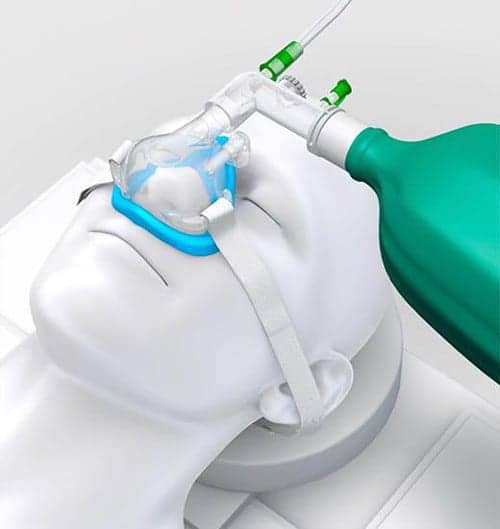
Vyaire’s SuperNO2VA anesthesia nasal mask.

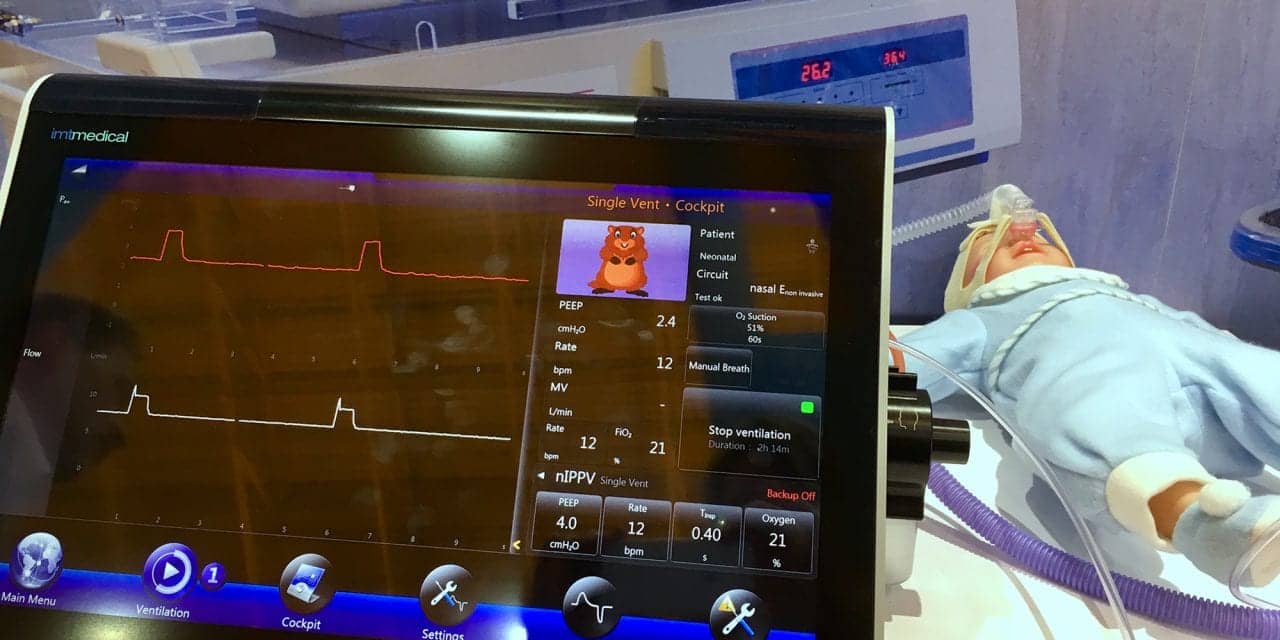








Can it be used for prolong ventilation like maquet servo-i & GE engstrom does?