Monaghan Medical Corp’s Aerobika Oscillating Positive Expiratory Pressure (OPEP) device has received a 2017 Innovative Technology designation from Vizient Inc. The designation was based on direct feedback from hospital experts who interacted with the Aerobika OPEP device at the Vizient Innovative Technology Exchange in Denver on Sept. 14, 2017.
“We are extremely pleased to have been awarded this prestigious recognition from Vizient,” said Dominic Coppolo, MBA, RRT, FAARC, vice president Clinical Strategy and Development. “We are the only OPEP device to have received this designation at this year’s Innovative Technology Exchange, and are proud it was recognized to deliver improved outcomes and enhanced safety.”
Aerobika is a drug-free, handheld mechanical oscillating positive expiratory pressure device that has been designed to address the structural and functional challenges in the airways of patients with COPD. It has been shown to improve lung function, exercise capacity and quality of life in COPD patients, and a real-world study showed that the device reduced exacerbation rates in patients during the critical 30-day post-exacerbation period, according to Monaghan.
These improved outcomes (equivalent to 6 fewer exacerbations per 100 patients per year) equate to a cost savings of $553 per patient with Aerobika compared with no OPEP/PEP use.
Vizient represents a diverse membership base that includes academic medical centers, pediatric facilities, community hospitals, integrated health delivery networks and non-acute health care providers and represents more than $100 billion in annual purchasing volume. Through its Innovative Technology Program, Vizient works with member-led councils and task forces to review potentially innovative products. If it is determined that a product is innovative, Vizient may award a contract outside of the competitive bid cycle.

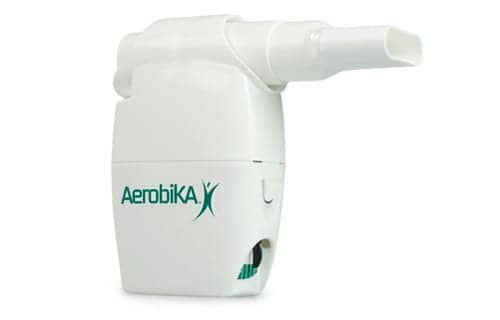





Good work to Trudell Medical Int. for developing this wonderful product.