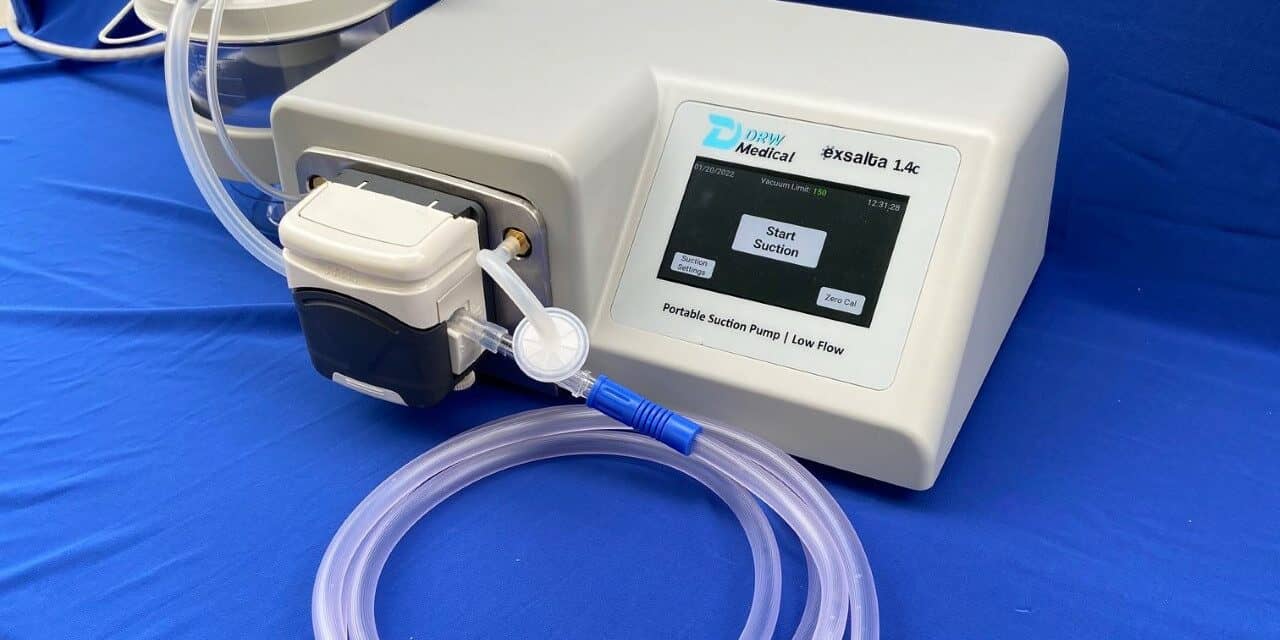
The US FDA has cleared DRW Medical’s EXSALTA secretion clearance device (EXtract Secretions And Leave The Air) for use on patients as indicated.
EXSALTA is the first peristaltic suction device for clearing endotracheal tube secretions for patients on ventilators. Its exceptional low flow technology, available at any suction setting, reduces risk of negative pressures in the lungs to help prevent alveolar collapse and hypoxia. EXSALTA is a smart secretion clearance solution for patients in whom the standard wall suctioning procedure may create adverse events.
The EXSALTA is a microprocessor-controlled tabletop suction device that offers clinicians smart technology to help mitigate adverse events associated with standard suctioning techniques.
The device uses peristaltic action to move fluids from the patient to a collection canister at a fixed flow rate of 1.4 L/min. The user selects a desired vacuum level that is independent of the flow rate.
EXSALTA was built to gain control over the rate at which secretions and air are evacuated from the patient’s lungs. It is known that suctioning a patient’s airways is associated with adverse events, such as hypoxemia, atelectasis, bradycardia, tachycardia, and airway mucosal damage. It is hypothesized that the severity of adverse events are increased due to excessive evacuation of air from the patient’s lungs during removal of secretions.