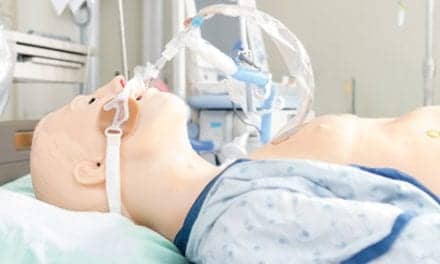
Philips Respironics Inc is recalling its V60 ventilators and V60 plus ventilators, according to a notice on the US Food and Drug Administration’s (FDA) website. The ventilators contain power management printed circuit board assemblies that were distributed by a third-party warehouse and do not meet ventilator standards, per the notice.
The FDA has identified this as a Class I recall, the most serious type of recall. Use of these devices may cause serious injuries or death. This recall is a voluntary correction, not a product removal.
Philips Respironics V60 ventilators and V60 plus ventilators are used to provide breathing assistance to pediatric and adult patients. A ventilator is intended to mechanically control or assist patient breathing by delivering a predetermined percentage of oxygen. V60 ventilators and V60 plus ventilators are intended to be used inside hospitals.
The V60 ventilators and V60 plus ventilators are not always used in intensive care units where patients are directly monitored. There may be greater risk for patients who are not directly monitored in other hospital settings.
The use of the affected ventilators may cause serious adverse health consequences, including power failure, loss of ventilator function, and death. There have been no reports of death or injuries related to this recall.
Recalled product:
- Product names: Philips Respironics V60 ventilators and V60 plus ventilators
- Product codes: See recall database entry
- Distribution dates: May 6 to May 6
- Devices recalled in the US: Eight
- Date initiated by firm: Aug 25
On Aug 25, Philips sent all affected customers an Urgent Medical Device Correction letter. The letter requested customers to discontinue use of the devices and quarantine them. A Philips Respironics representative will contact the customers. The representative will provide instructions to remove or replace the Power Management PCBAs in the affected devices.