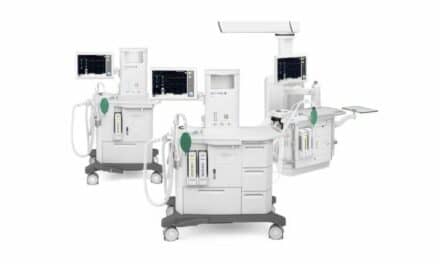
Michigan Instruments Inc (MII) will release an upgraded version of its Training and Test Lung, known as the “Michigan Lung,” in July 2014, according to the company. The upgrades include a new design as well as a new software interface, the PneuView3.
The combination of the new software application and improved product design brings significantly improved user capabilities, according to Michigan Instruments.
Developed 40 years ago, the Michigan Lung is a mechanical “respiratory simulation” of the human pulmonary system. It is capable of measuring airway pressure, lung pressure, tidal volume and 36 other parameters. The system is comprised of a precisely engineered mechanical test lung (or lungs), a set of electronic sensors, a signal conditioning package, and an integrated micro control unit. The PneuView3 software application calculates and displays, in real time, numerous respiratory parameters and waveforms. This data can also be exported for later review.
The latest comprehensive, fully-to-scale mechanical design allows for simulation of hundreds of patient scenarios, according to MII.
“Most test lungs on the market are built to perform a handful of simulations and are not fully to scale (parameters are adjusted according to a scale model). The Michigan Lung and PneuView3 System will provide users with a more comprehensive simulation to improve their respiratory products, care and treatments,” said Joe Baldwin, president of Michigan Instruments.
MII has designed and manufactured specialized medical equipment related to the fields of cardiovascular medicine, cardiopulmonary resuscitation, and respiratory therapy for over forty years. The company complies with FDA and International Organization of Standardization (ISO) regulations for Good Manufacturing Practices. More information is available at its website.