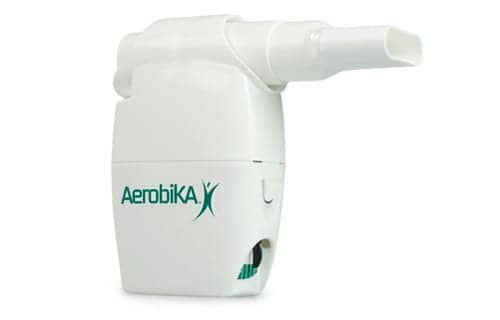
A recent study by Western University researchers, published in the Journal of Chronic Obstructive Pulmonary Disease, reveals that the use of the Aerobika device has a significant impact on quality of life for study patients suffering from chronic bronchitis. These study results show significant promise for millions of people suffering from COPD worldwide, according to a press release from Trudell Medical International, an affiliate of Monaghan Medical Corp.
The Aerobika device was evaluated in patients with a clinical diagnosis of COPD, conducted at The Robarts Research Institute, affiliated with the Schulich School of Medicine, Western University, London Canada. The findings indicated that for those sputum producing patients, daily use of the Aerobika device for three to four weeks produced significant improvements in clinical and patient reported outcomes. These included improved lung capacity, improved ease in mucus clearance, increased exercise tolerance and improvement in quality of life – significant results for COPD researchers and patients.
“Chronic Obstructive Pulmonary Disease (COPD) patients with chronic sputum production have worse clinical outcomes and accelerated lung function decline. Currently, there are few therapies available to support these patients and the results of this study are promising. After three or four weeks of using the Aerobika device daily, for COPD patients with chronic sputum production there was an improvement in how easily sputum could be coughed up and an improvement in quality-of-life,” says Dr Sarah Svenningsen, lead author on The Robarts Research Institute study.
“Patients suffering from COPD now have a safe and easy-to-use method to address the unmet need of mucus clearance, which can potentially reduce the need for hospitalization,” says Dominic Coppolo , Vice President of Clinical Affairs at Monaghan Medical Corporation. “Since its introduction to the market, the Aerobika device has already achieved a high degree of acceptance within the COPD patient population in the US.”
Dr. Jason Suggett, Group Director Science & Technology for Trudell Medical International concurs. “This research addresses a real and pressing concern in the treatment of COPD. Patients often take multiple medications for various conditions and medication adherence can be low. The Robarts Research Institute team’s study is an important validation of the positive patient outcomes experienced through this novel drug-free treatment.”
Additional quality of life evidence will be presented at the American Thoracic Society Conference in San Francisco on May 18, 2016.