[Updated 02.20.19 with revised comments from Gerard Criner, MD.]
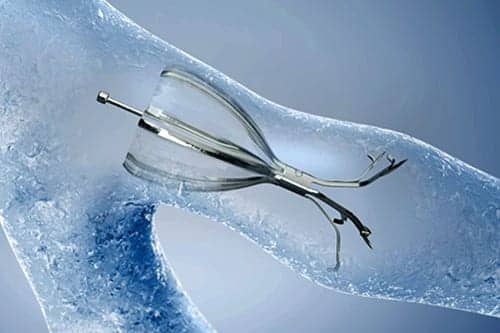
The Olympus Spiration Valve System (SVS), an FDA-approved endobronchial valve, is now commercially available for the treatment of severe emphysema.
Prominent guidelines now recommend minimally invasive bronchoscopic lung volume reduction using endobronchial valves as an alternative treatment option for severe emphysema to more invasive options, such as surgery, according to the company.
Placed in targeted airways of the lung during a short bronchoscopic procedure, the Spiration Valve is an umbrella shaped device that improves breathing by redirecting air from diseased parts of the lungs to healthier parts, enabling healthier tissue to expand.
SVS therapy may lead to volume reduction in the treated part of the lung, allowing the healthier tissue in the remaining portion of the lung to function more effectively, according to Olympus.
FDA approval of the SVS was based on results of the EMPROVE clinical trial demonstrating that patients treated with the SVS benefited from statistically significant and clinically meaningful improvements in lung function and quality of life compared to standard of care medical management. The results showed that the SVS offers a favorable risk benefit profile, with a short procedure time, Olympus reports.
“The EMPROVE trial corroborates and substantiates other data that shows how important it is to recognize that hyperinflation has a negative outcome on patients’ physiologic function, their quality of life, and even their survival,” said Gerard Criner, MD, Professor and founding chair of the Department of Thoracic Medicine and Surgery at the Lewis Katz School of Medicine, Temple University. “By having a potent therapy that significantly reduces lung hyperinflation, and is durable over time, you can start to see patients making truly meaningful improvements and achieving clinical outcomes such as improvements in lung function and quality of life.”
“The SVS offers significant benefit to a certain subpopulation of patients with emphysema,” said Kirk Voelker, MD, Interventional Pulmonologist and Director of Clinical Research at Sarasota Memorial Hospital. “There were a low number of complications, such as pneumothorax and pneumonia, which are all easily treated, but, importantly, we saw the durable effects of the therapy lasting through the follow-up year with significant improvement in quality of life.”
The SVS is approved for use in the treatment of emphysema in the US, EU, Australia, and New Zealand.








This sounds interesting. Is this covered by insurance? Will you send more info to my email address? T
Thanks,
Sharon
Do you know if this is MRI safe implant? Has this been tested in a magnetic field? What is it made of?
What stage must you be at to qualify for this procedure
We did this in the cath lab back in the 90s for patients with lung cancer and could see the benefit immediately. The patient immediately had increased volumes and ease of breathing. I was amazed then and now on how far this has come.
Where is this being performed?
What a god send! I’m calling my pulmonologist first thing Monday!! This is the most exciting news I’ve had for 8 years.
How deep into the lungs can this be used?
I would love more information!!! How can we get this? Is it covered by ins? Is it performed locally? Anything you can tell me would be wonderful
This is a second chance at life. I went through it and I am doing so so so much better. Can now live my life and enjoy every moment of it.