Olympus Corp has announced a voluntary field corrective action to address complaints of endobronchial combustion occurring during therapeutic procedures involving use of high-frequency therapy equipment in an oxygenated environment.
Olympus assessed the issue after receiving complaints of adverse events involving serious patient injury, where patients may suffer burns to the airway or lungs that may result in the need for additional medical intervention, prolonged procedure times, extended hospitalization, ICU care, and death.
Olympus took action to notify users about these complaints and reinforce existing warnings for use of bronchoscopes with high-frequency therapy equipment. This is not a removal action. Olympus bronchoscopes compatible for use with specified high-frequency therapy equipment can continue to be used following the instructions and warnings contained in the Olympus Operation Manual.
Olympus bronchoscopes are intended for use in endoscopic diagnosis and treatment within the airways and the tracheobronchial tree. A total of 28 BF series endoscope models are globally included in this action. Fifteen of those models were distributed in the US. The US Food and Drug Administration (FDA) and other regulatory bodies were notified of this action, according to a release form Olympus.
Olympus notified customers in the US by letter on Oct 12, requesting that healthcare personnel treating patients using an Olympus bronchofiberscope or bronchovideoscope carefully read the full Medical Device Corrective Action and ensure all personnel are completely knowledgeable and thoroughly aware of the warnings for use with compatible high-frequency therapy equipment, such as electrosurgical generators.
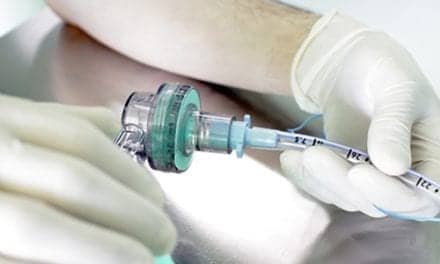
Olympus is reminding users to pay careful attention to warnings in the Operation Manual on high-frequency cauterization with Olympus bronchoscopes. Olympus Operation Manuals warn not to perform high-frequency cauterization while supplying oxygen. This may result in combustion during cauterization.
In addition, to avoid patient injury (burns, bleeding, and perforation) or damage to the device, confirm the electrode section of the electrosurgical accessory is at an appropriate distance from the endoscope’s distal end. Users may continue to use the device according to the existing instructions and warnings contained in the Operation Manual.
This is a separate field corrective action from one issued earlier this year regarding bronchoscope use with laser therapy equipment.
Adverse reactions or quality problems experienced with the use of these products may be reported to the FDA’s MedWatch program online.
For information or to report a problem, contact the Olympus Technical Assistance Center at 800-848-9024, Option 1, or email [email protected].