
A diagnosis of asthma has implications in several occupations, including the military, and an accurate assessment is essential based on the long-term consequences of the diagnosis. Bronchial provocation testing (BPT) is used in the evaluation and diagnosis of asthma; its primary goal is to identify and quantify airway hyperresponsiveness (AHR) to a specific stimulant.
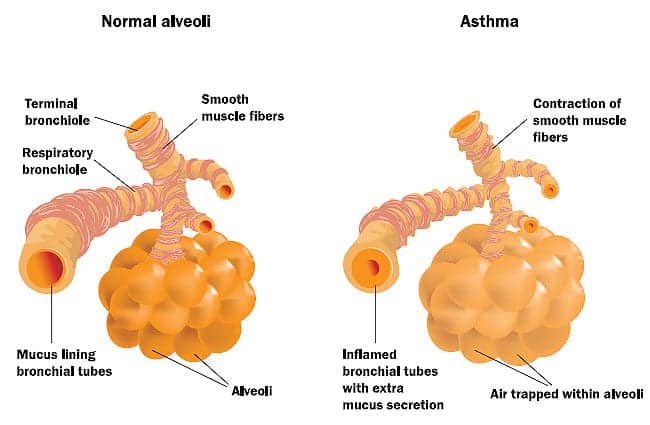
Asthma control is assessed using both components of current clinical control—symptoms, reliever use, and lung function—as well as future risk—exacerbations and decline in lung function. Taylor et al have defined asthma control as the degree to which the manifestations of asthma have been removed by treatment.1 Bronchial hyperresponsiveness (BHR) and airway inflammation are the two primary pathophysiologic manifestations of asthma. Physical signs and symptoms that are manifestations of asthma include cough, wheezing, chest tightness, and shortness of breath.
Direct Versus Indirect BPT Methods
Bronchoconstriction may be caused by either direct or indirect stimuli. Direct methods include histamine and methacholine. Both have been used for more than 30 years in clinical practice. Methacholine acts directly on the bronchial smooth muscle, causing it to contract. In the case of asthma, the AHR is increased, resulting in a smaller dose of the pharmacological agent and a lower provocative concentration to cause a 20% decrease in FEV1 (PC20).2 The relationship between direct stimuli and airway inflammation is inconsistent.3 There appear to be two components, airway inflammation and airway remodeling, that result in a positive response to a direct stimulus.4
In an indirect method, the stimuli cause a release of mediators such as histamine, prostaglandins, and leukotrienes, which in turn cause contraction of the bronchial smooth muscle.5 The smooth muscle response occurs as a consequence of existing inflammation. It may also be a result of activation of the cells or the stimuli as an irritant.2 Examples of indirect stimuli include exercise, dry powder mannitol, adenosine monophosphate, hypertonic saline, and hyperpnea of cold or dry air, also referred to as eucapnic voluntary hyperventilation (EVH) or isocapnic hyperventilation. The response to these indirect stimuli is related to airway inflammation and the concentration of the mediators released impacting the smooth muscle responsiveness. Indirect methods are useful to monitor the response to inhaled cortico-steroids; a positive response confirms currently active asthma.5
Direct Methods
Although methacholine and histamine are both direct methods, this section of the article will focus on methacholine because it is the only method approved in the United States for human use. It is important that the methacholine challenge test (MCT) is standardized so results are comparable between laboratories and to differentiate between normal and abnormal.6 American Thoracic Society (ATS) guidelines were published in 1999 for both exercise and methacholine challenge testing.7 These guidelines are currently being updated by a joint ATS/European Respiratory Society (ERS) committee. The guideline describes two separate methods for delivering the methacholine. One method is the 2-minute tidal volume breathing technique developed by Cockcroft et al.8 An individual inhales aerosolized methacholine from a jet nebulizer while tidal volume breathing for a 2-minute interval. The output must be calibrated to 0.13 mL/min.
The second method from Chai et al9 requires inhalation of five deep breaths to total lung capacity followed by a 5-second breath hold after each inhalation, using a dosimeter to deliver the methacholine. The nebulizer should be calibrated to deliver 9 µL per each actuation. The remainder of the procedure for testing is similar regardless of how the medication is delivered. Saline may be used as a diluent for baseline control. The methacholine dose is doubled from 0.03 mg/mL to 16 mg/mL. A total of 10 doses is given if the entire procedure is finished without a positive response. A 5-minute window between doses is allowed. The FEV1 is measured after each dose at 30 and 90 seconds. The highest acceptable FEV1 at each dose is selected. The decrease in FEV1 is calculated using the FEV1 obtained after the methacholine at the highest dose and the FEV1 post-diluent. The test is terminated after a 20% decrease in FEV1 occurs or the highest concentration of the drug has been delivered.
The ATS document also describes a quadrupling dose protocol. The ATS recommendations do address the possibility of using FEV1 as the only outcome measure. If this is the case, the expiratory time may be shortened. Caution should be used in the inspiration preceding the expiratory phase. A full inspiration must be achieved or the FEV1 will be falsely low; and, without a complete expiration to residual volume, it is difficult to evaluate the quality of the inspiratory phase of the test.7
Although initial studies indicated that the methods were similar in results, newer evidence suggests that the 2-minute tidal volume breathing technique results in a consistently lower PC20, specifically in individuals with mild AHR. The methods appear to be more comparable in individuals with moderate to greater AHR. Cockcroft cites two reasons for this disparity: The tidal volume technique delivers a larger dose of methacholine, while the dosimeter technique results in bronchodilatation with the five maximal inhalations and a bronchoprotection effect.6
Quality control of the nebulizers is an important consideration because the actual output has a significant impact on the results of the test. Because nebulizer output can vary based on model, unit, and usage, the output should be checked initially and at regular intervals using the ATS recommendations.7
Methacholine has a high negative predictive power, which means current asthma is generally ruled out with a PC20 greater than 16 mg/mL. The positive predictive power is high and specificity is high when the PC20 is less than 1 mg/mL. At this level, a methacholine challenge is similar to indirect challenges.6
Indirect Methods
The indirect methods commonly performed in the pulmonary function laboratory have historically included exercise, EVH, and hypertonic saline. Recently, dry-powder mannitol has been approved as another indirect method in the United States. In 2007, the Global Initiative for Asthma Guidelines (GINA) added exercise and mannitol as indirect challenges to establish a diagnosis of asthma. The ATS described a standardized method for performance of exercise-induced bronchoprovocation (EIB) studies in 1999. This was the first indirect method to be standardized. Exercise intensity is selected to produce 4 to 6 minutes at near maximum targets with a total exercise time of 6 to 8 minutes in a dry environment. Heart rate or ventilation can be used to determine the maximum target level for the exercise. FEV1 is measured at baseline and 5, 10, 15, 20, and 30 minutes post-exercise. The highest FEV1 at each level is selected. A decrease of 10% to 15% in the FEV1 from baseline to post-exercise is considered a positive response.7 EIB studies have remained important to assess the severity and control of asthma.
Eucapnic voluntary hyperventilation was initially developed as a method to diagnosis asthma in army recruits and EIB in elite athletes. For this method, an individual breathes dry air for 6 minutes at a ventilator target of 30 times the FEV1. Severe decreases in FEV1 can occur, so the method is used in a select population. A decrease of 10% in the FEV1 has been suggested for a positive test. Similar to exercise testing, treatment with inhaled corticosteroids (ICS) decreases the responses to EVH.10
A third indirect method using hypertonic saline (4.5% NaCl) has an advantage over EIB and EVH because sputum can also be collected. Both the number of eosinophils present and the mast cells in the epithelium can be evaluated. A high-output ultrasonic nebulizer is used to generate the aerosol at increasing time intervals (0.5, 1, 2, 4, 8 min). Sensitivity to hypertonic saline is decreased after treatment with ICS. A positive test is considered a 15% to 20% decrease in FEV1. The same mediators measured with exercise are also present with hypertonic saline. These mediators are released from the mast cell and include such substances as prostaglandin, leukotriene, and histamine.10
After experience with hypertonic saline, the mannitol challenge was developed. Mannitol is an osmotic agent considered safe in both adults and children. It increases osmolarity of the airway surface liquid. Mannitol is useful to aid in the diagnosis of asthma, and a positive test is consistent with inflammation and the associated mediators. It is also helpful in the ongoing assessment of active airway inflammation. The provocation agent is encapsulated and delivered by a special dry-powder inhaler in increasing doses (0, 5, 10, 20, 40, 80, 160, 160, 160 mg). The FEV1 is measured 1 minute after each dose. The challenge test time is critical, and prolonged time between doses may impact the results.10 There are contraindications to a BPT with mannitol: known hypersensitivity to the mannitol or capsule ingredients, airflow limitation (reduced FEV1/FVC) and FEV1 less than 50% of the predicted value or 1.0 liter, or the inability to perform acceptable and repeatable baseline spirometry. Equipment and supplies required include the mannitol kit, timer, calculator, bronchodilator, calibrated spirometer, and appropriate emergency equipment. A positive test to mannitol is reported as a 15% decrease in FEV1 at a total cumulative dose of 635 mg or less or a 10% decrease in FEV1 between consecutive doses.11,12 The PD15 expresses the sensitivity. Reactivity is the response-dose ratio calculated as the final percent fall in FEV1 divided by the total cumulative dose to induce that percent fall in FEV1. As an indirect stimulus, it is a surrogate for exercise, EVH, and hypertonic saline.10
The failure rate for laboratory exercise protocols is fairly high compared to EIB in the field. This has prompted the evaluation of mannitol and EVH as potential surrogates.13 Exercise-induced bronchoprovocation is an important marker as it is common in individuals with active asthma. Additional research is needed to determine if other tests using indirect stimuli are an economical and better asthma management tool than exercise.5
Conclusion
The use of bronchial provocation testing is important to identifying asthma in individuals with what appears to be normal lung function during the initial spirometry test who will benefit from a therapeutic intervention such as inhaled corticosteroids. It is important to establish a correct diagnosis and exclude those who do not have asthma even when symptoms may suggest asthma. Diseases such as COPD and allergic rhinitis may also include hyperresponsiveness.
Asthma is a complex disease that may require more than one BPT method, direct and indirect, to confirm or exclude the diagnosis of the disease. Approximately 30% of individuals with mild EIB are not positive to mannitol or methacholine.10 As the research continues to clarify the sensitivity and specificity of each method, our understanding of the most appropriate method to select in specific populations will increase. Method comparison in multiple populations, levels of baseline lung function, and severity of asthma are required to improve our understanding. The body of scientific research in this area will continue to expand with the addition of mannitol as another approved agent.
The challenge is to carefully investigate each method and its relationship to an initial physician diagnosis of asthma, a previous diagnosis, or the length of a current diagnosis and benefits of a therapeutic intervention on AHR. Understanding the mechanisms of increased airway reactivity will likely continue to challenge the scientific community for many years.
Susan Blonshine, RRT, RPFT, AE-C, FAARC, is president and CEO of TechEd, a respiratory care consulting service in Mason, Mich. For further information, contact [email protected]
References
- Taylor DR, Bateman ED, Boulet LP, et al. A new perspective on concepts of asthma severity and control. Eur Respir J. 2008;32:545–54.
- Busse WW. The relationship of airway hyperresponsiveness and airway inflammation. Airway hyperresponsiveness in asthma: its measurement and clinical significance. Chest. 2010;138(2 Suppl):4S–10S.
- Crimi E, Spanevello A, Neri M, Ind PW, Rossi GA, Brusasco V. Dissociation between airway inflammation and airway hyperresponsiveness in allergic asthma. Am J Respir Crit Care Med. 1998;157:4–9.
- Cockcroft D, Davis B. Direct and indirect challenges in the clinical assessment of asthma. Ann Allergy Asthma Immunol. 2009;103:363-72, 400.
- Brannan JD. Bronchial hyperresponsiveness in the assessment of asthma control. Airway hyperresponsiveness in asthma: its measurement and clinical significance. Chest. 2010;138(2 Suppl):11S–17S.
- Cockcroft DW. Direct challenge tests. Airway hyperresponsiveness in asthma: its measurement and clinical significance. Chest. 2010;138(2 Suppl):18S–24S.
- Crapo RO, Casaburi R, Coates AL, et al. Guidelines for methacholine and exercise challenge testing-1999. This official statement of the American Thoracic Society was adopted by the ATS Board of Directors, July 1999. Am J Respir Crit Care Med. 2000;161:309-329.
- Cockcroft DW, Killian DN, Mellon JJA, Hargreave FE. Bronchial reactivity to inhaled histamine: a method and clinical survey. Clin Allerg. 1977;7:235-43.
- Chai H, Farr RS, Froehlich LA, et al. Standardization of bronchial inhalation challenge procedures. J Allergy Clin Immunol. 1975;56:323–7.
- Anderson SD. Indirect challenge tests. Airway hyyperresponsiveness in asthma: its measurement and clinical significance. Chest. 2010;138(2 Suppl):25S–30S.
- Anderson SD, Charlton B, Weiler JM, et al. Comparison of mannitol and methacholine to predict exercise-induced bronchoconstriction and a clinical diagnosis of asthma. Respir Res. 2009;10:4.
- Brannan JD, Anderson SD, Perry CP, et al. The safety and efficacy of inhaled dry powder mannitol as a bronchial provocation test for airway hyperresponsiveness: a phase 3 comparison study with hypertonic (4.5%) saline. Respir Res. 2005;6:144.
- Rundell KW, Anderson SD, Spiering BA, Judelson DA. Field exercise vs. laboratory eucapnic voluntary hyperventilation to identify airway hyperresponsiveness in elite cold weather athletes. Chest. 2004;125:909–15.








