Congestive heart failure (CHF) is the term used to describe the physiological state in which cardiac output is insufficient. It is the fastest growing cardiovascular disorder in the United States, one of only a few increasing in incidence and prevalence.1 As the population ages, the incidence of congestive heart failure is rising dramatically; it is estimated that CHF affects about 10 per 1,000 people age 65 and older.
In the United States, approximately 6 million people suffer from heart failure, with an additional 670,000 new cases of heart failure diagnosed each year.2 Heart failure is the most common reason for hospitalization in the elderly and carries an annual cost of approximately $39 billion.2 In the Medicare population, heart failure is also the most common reason for hospital readmission within the sensitive period of 30 days following discharge.3 This is of growing importance as we enter a new era of clinical accountability, one in which a major health care payor such as Medicare imposes financial penalties against hospitals with a high incidence of readmissions.
Congestive heart failure sits at the top of a list of chronic, progressive diseases that produce significant burdens on patients, families, and the health care system. In addition to the obvious financial burdens, there are significant quality-of-life issues faced by patients with CHF, along with their families and caregivers. Like many other chronic and progressive diseases, in CHF it is essential that individuals at risk be diagnosed and treated early.
Disease Classification and Treatment
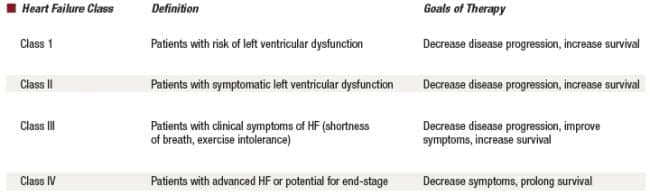
Congestive heart failure is a progressive heart disease caused by myriad factors, including coronary artery disease, hypertension, congenital heart disease, chronic arrhythmias, and chronic obstructive lung disease. Like other chronic, progressive disorders, once diagnosed, CHF is classified by the level of cardiac impairment. The left, right, or both sides of the heart can be affected. Right-sided heart failure (cor pulmonale) results in ineffective blood flow into the pulmonary vasculature, and left-sided heart failure results in ineffective blood flow to the body. The Table lists the four heart failure classes, definitions, and basic treatment goals.
The disorder is most often medically managed with myriad drug therapies aimed at improving cardiac performance, slowing progression, and reducing symptoms. Common drug therapies include, but are not limited to, beta blockers, angiotensin converting enzyme (ACE) inhibitors, angiotensin receptor blockers (ARB), various diuretics, selective vasodilators, calcium channel blockers, and anticoagulants. As the disease progresses, chronic treatments can become more comprehensive and include more drug combinations, supplemental oxygen therapy, and both noninvasive and invasive ventilation support. Because most of the severe cardiopulmonary symptoms do not present until the later stages, early assessment and treatment is often overlooked.
Table.
Cheyne-Stokes Respiration in CHF
Cheyne-Stokes respiration (CSR) is an abnormal respiratory pattern associated with CHF, and, while many clinicians are familiar with CSR, particularly in end-stage cardiopulmonary disease, mild CSR may be present in many stable, early-stage asymptomatic patients.
Cheyne-Stokes respiration is characterized by a crescendo-decrescendo alteration in tidal volume that is separated by periods of apnea or hypopnea. The crescendo-decrescendo breathing alterations in CSR compensate for the changing serum partial pressures of oxygen and carbon dioxide that result from sluggish circulation with timing delays between the pulmonary artery and central and peripheral chemoreceptors. Up-regulation in peripheral chemoreceptor sensitivity results in rapid compensatory changes in respiratory drive in response to small changes in serum oxygen and carbon dioxide. Although the carbon dioxide levels rise and fall in relation to changes in minute volume, the mean CO2 level remains below normal.
Hypoxia clearly plays a central role in the initiation of CSR, as evidenced by the attenuation of periodic breathing following administration of supplemental oxygen. Gottlieb and colleagues studied hemodynamic stress associated with heart failure and concluded that prevention of hypoxia is especially important in this at-risk population of patients.4
Diagnosing Nocturnal Hypoxemia
Patient complaints that may be suggestive of nocturnal hypoxemia may be easily confused with other comorbid symptoms, and, therefore, a typical outpatient exam is not adequate to detect sleep-related symptoms. Resting and ambulating blood oxygen levels are not indicative oxygen levels during sleep. Nocturnal desaturation is directly correlated to sleep disturbance and CSR in stable CHF patients with treated heart failure and no daytime evidence of hypoxemia.5

Diagnosing nocturnal hypoxemia is relatively simple and inexpensive, and it can be easily performed in the patient’s home. Although complete polysomnography will identify numerous sleep disturbances and should be used if more serious sleep disorders are suspected, the use of a single, overnight pulse oximetry study can serve as an adequate diagnostic tool for identifying mild CSR and hypoxemia in patients with CHF.5 Not all clinicians agree on the definition of nocturnal hypoxemia, although the Medicare coverage policy for nocturnal home oxygen therapy6 serves as a standard of practice: “An arterial PO2 at or below 55 mm Hg, or an arterial oxygen saturation at or below 88 percent, for at least 5 [cumulative] minutes taken during sleep for a patient who demonstrates an arterial PO2 at or above 56 mm Hg or an arterial oxygen saturation at or above 89 percent while awake.”
Evidence-Based Treatment
Oxygen therapy and continuous positive airway pressure (CPAP) are the common treatment options for stable CHF patients with documented sleep disturbances, CSR, and hypoxemia. There are numerous studies demonstrating the efficacy of low flow nasal oxygen therapy as a treatment for CSR in patients with stable CHF, including studies that compare nocturnal oxygen therapy (NOT) to CPAP.
- In a randomized, controlled study, Sasayama and colleagues studied the effects of nocturnal oxygen therapy on stable CHF patients with CSR and nocturnal hypoxemia and concluded NOT improves sleep-disordered breathing (SDB), left ventricular function, and QOL.7
- Shigemitsu et al8 studied the use of NOT in a population of CHF patients diagnosed with central SDB (apnea/hypopnea index =20) and hypoxemia. They concluded NOT significantly decreased the apnea/hypopnea index (AHI) and suggested that NOT may prevent the progression of CHF in patients with SDB and hypoxemia.
- Javaheri and colleagues9 studied a group of stable CHF patients with polysomnographic evidence of periodic breathing and hypoxemia to determine the clinical effects of nasal NOT and concluded that in patients with stable heart failure, nasal NOT significantly improves periodic breathing and virtually eliminates clinically significant desaturation.
- In a similar study, Staniforth and colleagues10 reported similar outcomes for breathing and arterial saturation, in addition to a reduction in urinary catecholamine levels.
- In a prospective, randomized controlled trial, Krachman and associates11 compared NOT with CPAP in patients with CHF and CSR and concluded NOT and nCPAP are equally effective in decreasing the AHI in CHF patients with CSR.
PAP therapies, in the forms of CPAP, bi-level, and, most recently, adaptive servo-ventilation (ASV), have been used to treat mild to severe CSR. Studies focused on the effectiveness of these therapies have met with mixed results, although in advanced CSR, there is a clear benefit, particularly for treatment with ASV. These mixed outcomes may, at least in part, be attributed to the targeting of therapy to treat symptoms (irregular breathing) rather than the cause (poor oxygenation) of those symptoms.
- In a controlled trial, Bradley and colleagues12 randomized 258 CHF patients with central apnea to receive CPAP or no CPAP and followed them for 2 years. They concluded that CPAP therapy improved central apnea, nocturnal desaturation, and left ventricular ejection fraction. CPAP also lowered norepinephrine levels.
- Buckle et al13 and Davies et al14 compared CPAP to room air and sham CPAP (respectively) in controlled randomized trials. Both studies found no difference in AHI, and Buckle et al found no improvement in nocturnal saturation or sleep efficiency.
- Krachman and colleagues11 compared the effect of oxygen therapy with nasal CPAP on CSR in patients with CHF. Polysomnography was performed on 25 stable CHF patients with 14 identified as having CSR. Patients identified as having CSR were randomized to a night of oxygen therapy and another night on nasal CPAP therapy. When compared with baseline data, both oxygen and nasal CPAP significantly decreased the AHI with no significant difference between the two modalities. The mean oxygen saturation increased significantly and to a similar extent with both oxygen and CPAP therapies.
- Arzt and Bradley15 evaluated studies that reviewed either oxygen therapy or CPAP treatment or compared both and concluded that reduction in AHI was similar in the two therapies, but greater improvement in oxygenation was achieved when low flow oxygen was the primary treatment.
Compliance to Therapy
The best, most effective therapy is of no value if patients fail to adhere to it. Successful, effective treatment of CSR is an issue of practical comfort and convenience for the patient. Buckle et al13 found that most of the CHF/CSR patients treated with CPAP did not tolerate the therapy and they would not have accepted long-term therapy even if it had shown some benefit to them. CPAP compliance has long been a concern, with data suggesting about 50% of CPAP users are noncompliant. Although these studies are primarily directed at the treatment of obstructive sleep apnea, they highlight issues such as air leaks, nasal and sinus discomfort, pressure sores, and others that drive patients away from adhering to therapy. Low flow oxygen therapy is much more familiar and less intrusive than PAP, particularly when it is administered as a nocturnal-only therapy. It is relatively easy for the patient to use and is not associated with the numerous side effects and patient complaints recognized in PAP therapies.
Summary
Stable, normoxic CHF patients may have undiagnosed CSR with associated nocturnal hypoxemia. Diagnosis and treatment are relatively simply and can be employed in the outpatient setting and in the patient’s home via overnight pulse oximetry. Patients with stable CHF demonstrating nocturnal desaturation may be ideal candidates for early treatment with low flow nasal oxygen therapy. As the disease progresses, additional and more comprehensive therapies, such as ASV, may be integrated into the treatment plan.
Joseph Lewarski, BS, RRT, FAARC, is vice president of clinical affairs, Invacare Corp, Elyria, Ohio. For further information, contact [email protected].
References
- Yamani M, Massie BM. Congestive heart failure: insights from epidemiology, implications for treatment. Mayo Clin Proc. 1993;68:1214-8
- Lloyd-Jones D, Adams RJ, Brown TM, et al. Heart disease and stroke statistics—2010 update: a report from the American Heart Association. Circulation. 2010;121:948-54.
- Jencks SF, Williams MV, Coleman EA. Rehospitalizations among patients in the Medicare fee-for-service program. N Engl J Med. 2009;360:1418-28.
- Gottlieb JD, Scwartz AR, Marshall J, et al. Hypoxia, not the frequency of sleep apnea, induces hemodynamic stress in patients with heart failure. J Am Coll Cardiol. 2009;54:1706-12.
- Staniforth AD, Kinnear WJM, Starling R, Crowely AJ. Nocturnal desaturation in patients with stable heart failure. Heart. 1998;79:394-9
- Centers for Medicare and Medicaid Services. National Coverage Determination (NCD) for Home Use of Oxygen (240.1). Available at: www.cms.gov/medicare-coverage-database/details/ncd-details.aspx?NCDId=169&ncdver=1&DocID=240.2&SearchType=Advanced&bc=IAAAABAAAAAA&. Accessed January 18, 2012.
- Sasayama S, Izumi T, Seino Y, Ueshima K, Asanoi H; CHF-HOT Study Group. Effects of nocturnal oxygen therapy on outcomes measures in patients with chronic heart failure and Cheyne-Stokes respiration. Circ J. 2006;70(1):1-7.
- Shigemitsu M, Nishio K, Kusuyama T, Itoh S, Konno N, Katigiri T. Nocturnal oxygen therapy prevents progress of congestive heart failure with central sleep apnea. Int J Cardiol. 2007;115:54-60.
- Javaheri S, Ahmed M, Parker TJ, Brown CR. Effects of nasal O2 on sleep-related disordered breathing in ambulatory patients with stable heart failure. Sleep. 1999;22:1101-6.
- Staniforth AD, Kinnear WJM, Starling R, Hetmanski DJ, Cowley AJ. Effect of oxygen on sleep quality, cognitive function and sympathetic activity in patients with chronic heart failure and Cheyne-Stokes respiration. Eur Heart J. 1998;19:922-8.
- Krachman SL, D’Alonzo GE, Berger TJ, Eisen HJ. Comparison of oxygen therapy with nasal continuous positive airway pressure on Cheyne-Stokes respiration during sleep in congestive heart failure. Chest. 1999;116:1550-7.
- Bradley TD, Logan AG, Kimoff RJ, et al. Continuous positive airway pressure for central sleep apnea and heart failure. N Engl J Med. 2005;353:2025-33.
- Buckle P, Millar T, Kryger M. The effect of short-term nasal CPAP on Cheyne-Stokes respiration in congestive heart failure. Chest. 1992;102:31-5.
- Davies RJ, Harrington KJ, Omerod OJ, Stradling JR. Nasal continuous positive airway pressure in chronic heart failure with sleep disordered breathing. Am Rev Respir Dis. 1993;147:630-4.
- Arzt M, Bradley TD. Treatment of sleep apnea in heart failure. Am J Respir Crit Care Med. 2006;173:1300-8.